Pipeline
You can scroll left and right to view the full content.
| Pipeline | Development Projects | Target diseases | Development of drug candidate | Preclinical trials | Safety test | Field trials | Product approval |
|---|---|---|---|---|---|---|---|
| vFT001 | FT005-002 (MyoLac II) |
Industrial animal improvements |
|
||||
| vFT005 | |||||||
| vFT011 | FT005-005 (Lobster) |
Melanoma, Lymphoma |
|
||||
| vFT012 | FT005-004 (Crab) |
Osteosarcoma, Mast cell tumors, Mammary tumors |
|
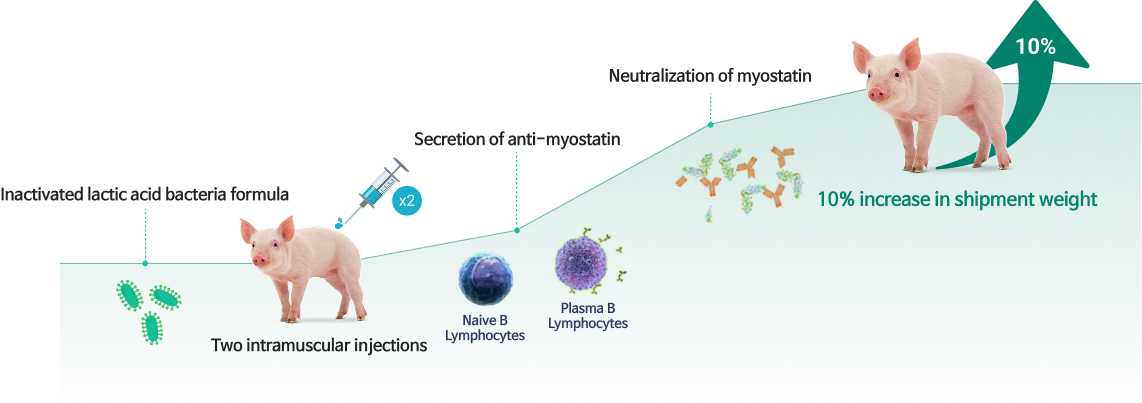
MyoLac II (vFT005): Injectable treatment for enhancing pig productivity
Future & Tech is currently developing MyoLac II, an injectable veterinary pharmaceutical for improving the productivity of industrial animals (including pigs) by utilizing its proprietary GOST technology.
Myostatin, a substance that inhibits muscle growth, is expressed in the muscle cells of all vertebrates, including industrial livestock and humans.
Among known therapeutic development targets for muscle disorders, myostatin and its receptor are the most actively researched in clinical trials by many global pharmaceutical companies.
MyoLac II is composed of inactivated lactic acid bacteria, where myostatin has been expressed as surface antigen protein. It is a candidate treatment based on GOST technology aimed at treating muscle disorders.
MyoLac II has been injected twice in experimental animals (mice) and target animals (pigs), which have confirmed for antibody formation against myostatin, a 10% increase in body weight, and therapy safety. MyoLac II is in the process of requesting product approval as an injectable veterinary pharmaceutical from the Animal and Plant Quarantine Agency of Korea.
Therapy mechanism of MyoLac II
[Target animal efficacy test (XP Bio test): 1-week-old pigs, two injections (6-month study)]
- Safety (Low, Medium, High dosage): administrated two intramuscular injections with concentrations of 1.0x108, 1.0x109, 1.0x1010 cells/head, where the control and treated groups showed no significant differences in mortality or generic symptoms
- Validation of efficacy: dosages of 1.0x108 and 1.0x109 cells/head resulted in average 13.5% and 10.8% weight gain, respectively.
- Formation of anti-myostatin antibody was confirmed in treated groups (antibody formation showed meaningful correlation with weight gain)
- Identified optimal dosage
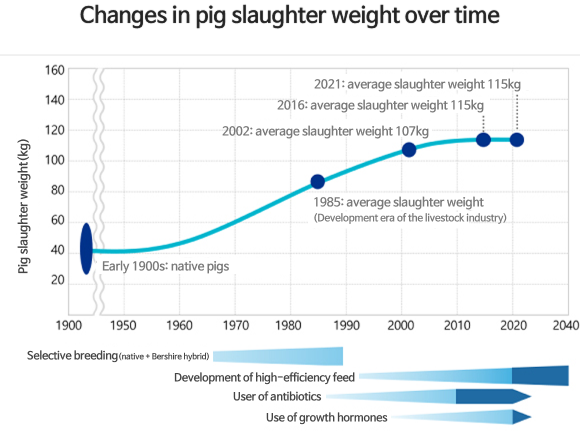
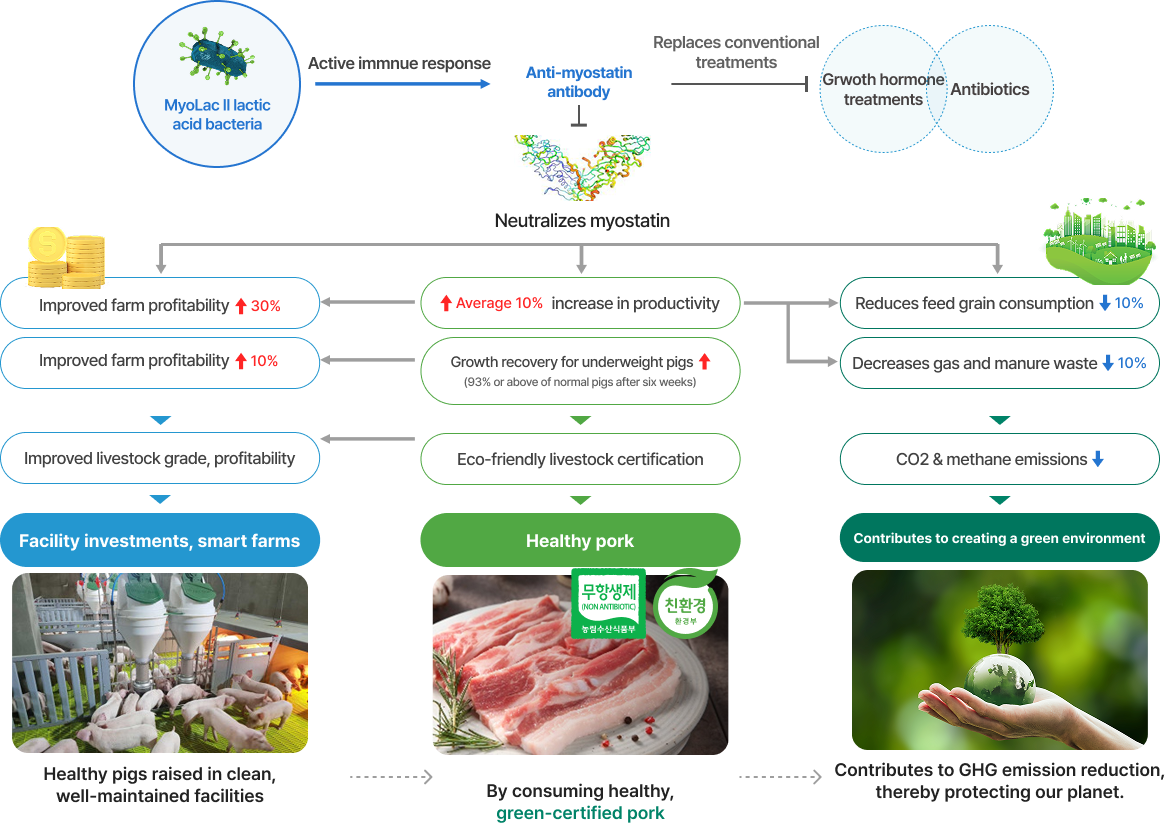
Background for the development of livestock enhancement pharmaceutical MyoLac II
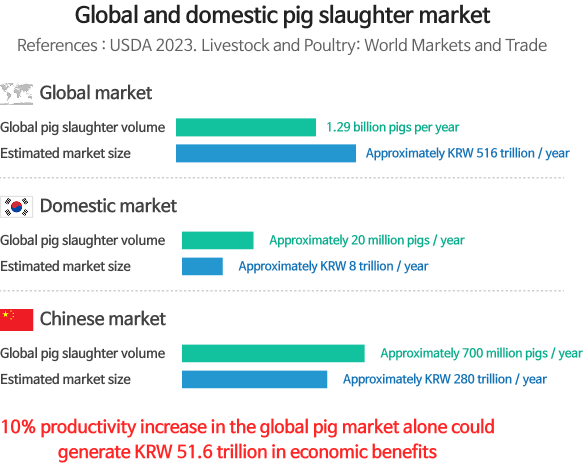
Expected benefits of developing MyoLac II seen through productivity uplift in pigs
-
- Enhanced productivity leads to a 30% increase in farm profitability (for an average domestic farm with 200 sows, net profit increases from KRW 320 million to KRW 440 million)
- Replaces pork imports [expected 26% reduction in annual pork import from 374,000 tons (additional domestic production of 106,000 tons with enhanced productivity)]
- Since the amino acid sequence for mature (active) myostatin are 98-100% identical in vertebrates, the technology could be extended across various livestock species to improve productivity.
- Normalizes and improves growth of underweight and weakened piglets (unmet medical needs remain to date), shortening time to market and enhancing productivity
-
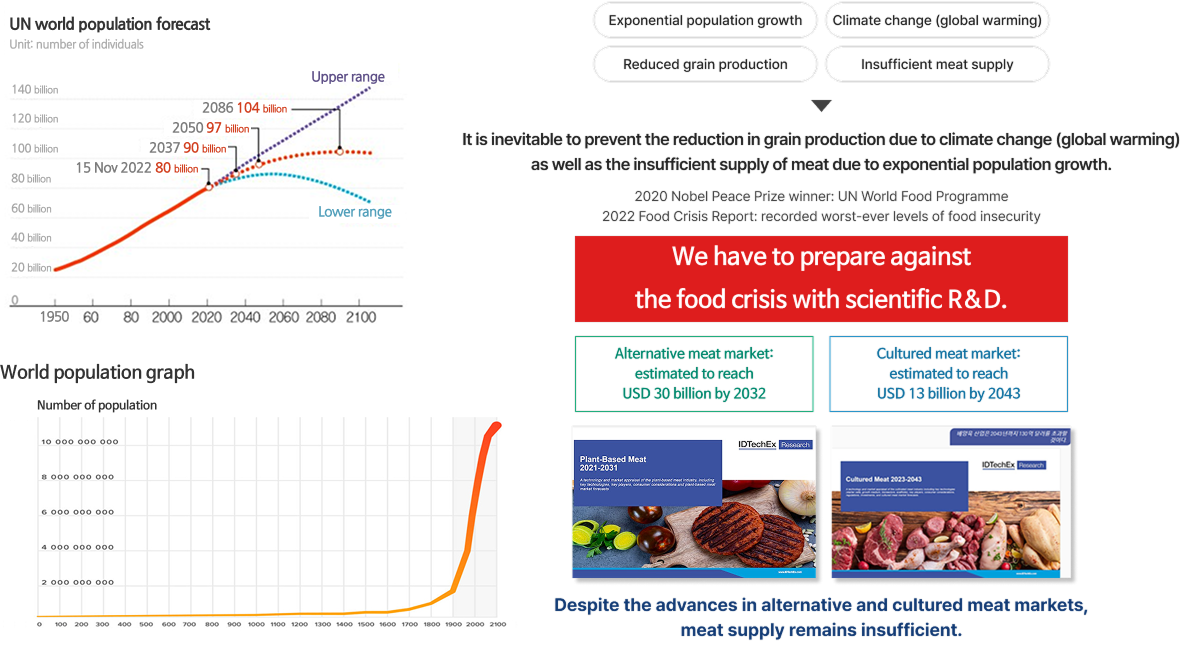
- Addresses global meat supply imbalance by improving the productivity of industrial animals
- Prevent the emergence of multiple antibiotic-resistant bacteria by replacing antibiotic feed additives that may leak out to the environment (such bacteria are expected to cause 2 million deaths globally by 2050)
- Provide safe meat to consumers by replacing growth hormone treatments, which are currently banned from use due to human safety concerns.
Market analysis and technology characeristics
Changes in governmental policies
- Regulations restricting the use of antibiotics and growth hormones for increased meat yield
- Regulations on the use of antibiotics
: Banned the use of antibiotic additives in compound feed from July 2011
: 2026, Global antibiotics market in livestock: 2021, USD 4.7 billion (CAGR: 3.6%), 2026 antibiotics market: USD 4.16 billion, 73.4% of total USD 5.6 billion (KRW 5.6 trillion) was used in livestock sector - Regulations on growth hormone treatments
: Designated as a risk element in terms of safety and efficacy
(Animal and Plant Quarantine Agency, Scope and tolerance thresholds of harmful substances in feed [related to Article 11, Paragraph 1])
: 2023, Global beta agonist feed additives market: USD 89 billion (KRW 120 trillion)
- Regulations on the use of antibiotics
- Introduction of eco-friendly livestock certification program (November 2020)
- There is urgent need for new pharmaceuticals certified as eco-friendly that can replace antibiotics and growth hormone treatments
Development benefits
Cookie notice
By clicking 'Accept all cookies', you agree to the storing of cookies on your device and to the associated processing of data to enhance site navigation, analyse site usage, and assist in our marketing and performance efforts.
Cookie Consent Settings
- Strictly Necessary Cookies
- Functional Cookies
- Marketing/Advertising Cookies
- Statistics Cookies